On May 29th the FDA granted Emergency Use Authorization for our rapid antibody test kits made by Healgen Scientific LLC.
While the FDA has approved many new tests for COVID-19 in the past months, only a handful of these rapid tests have achieved this status.
Among FDA EUA approved rapid antibody tests, the sensitivity and specificity was exceptional, as this independent study done by Erasmus reported [Orient Gene is the parent company of Healgen Scientific LLC].
An EUA from the FDA is based on Section 564 of the Federal Food, Drug and Cosmetic Act (FD&C) which provides a fast track process for introducing drugs and other medical products during an emergency if they meet certain criteria.
As made clear by the Erasmus study as well as this independent study , the Healgen Scientific IgG sensitivity scored well above the accepted range.
IgG is the post-infection antibody that may indicate immunity.
Keeping track of IgG positive patients will help shape public policy as we learn the full scope of the infection to produce more accurate models.
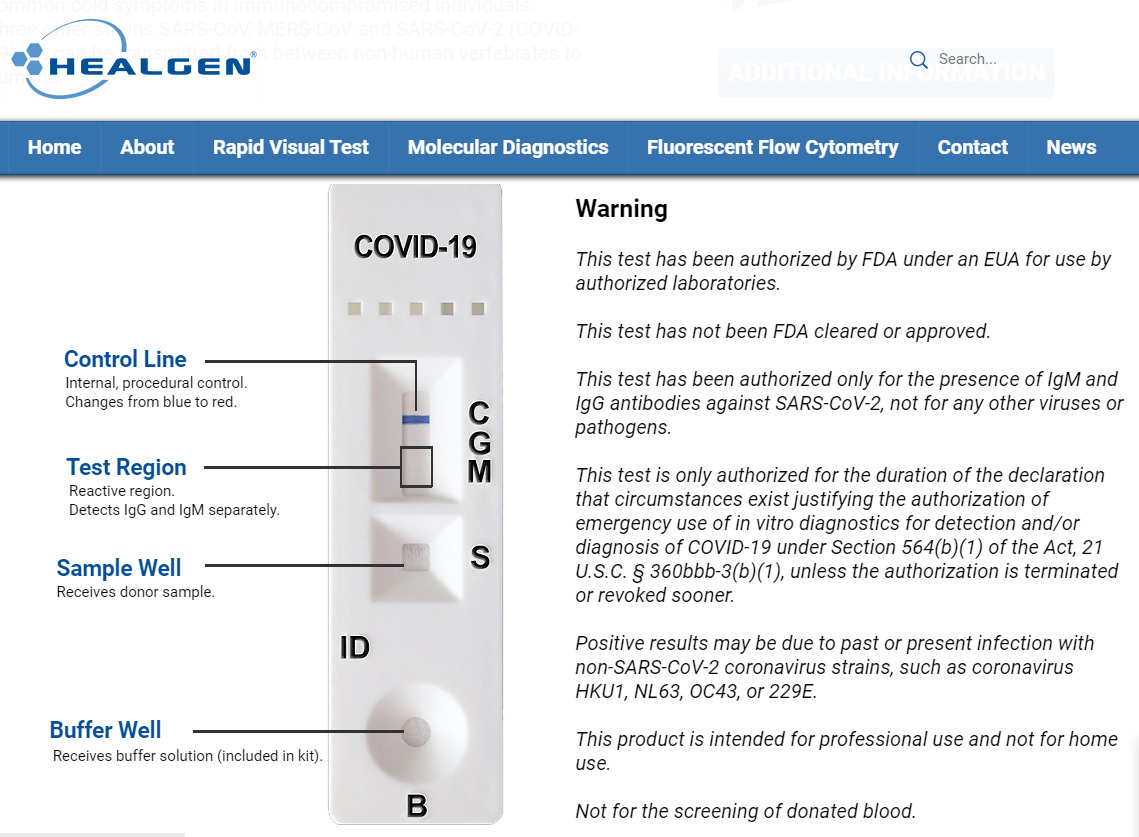
While there are no material changes to our IgM/IgG antibody test kits, the EUA does make an impact on how the test is done and what distributors must do to be compliant:
- The test must now be performed using venous whole blood drawn by a professional phlebotomist. The approval no longer recommends finger-prick draws for usage
- EUA approved antibody test kit boxes will now feature the FDA approval label.
- As a distributor of an FDA EUA approved antibody test, Stellar Scientific is required to publish and make available usage guidelines for health care professionals and for patients receiving the test.
- As a distributor for an FDA EUA approved antibody test, Stellar Scientific is required to keep records of authorized laboratories that purchase these tests and report to Healgen Scientific incidences of suspected false positives and false negatives.
- Clinical labs may collect reimbursement for these serology tests from insurance.
It is likely the price will increase, but we will be able to offer discounts for bulk purchases of 1000 antibody tests and above.


